The Federal Government, in its shipping and transportation standards, defines etiologic agents as microorganisms that cause disease in humans including the following: bacteria, bacterial toxins, viruses, fungi, rickettsia, protozoans, parasites and prions. These disease-causing microorganisms may also be referred to as infectious agents or infectious substances, and the materials, such as body fluids and tissues that contain them, are referred to as infectious materials. When a package of infectious material is being imported into the United States, it must have an importation permit approved by the CDC. Organisms such as mosquitoes that might transmit infectious diseases to other humans are called vectors. Vectors may require permits from agencies such as the CDC, USDA or the California Department of Public Health.
It is important to obtain a CDC permit PRIOR to requesting an etiologic specimen from a source outside the United States. The Stanford University Administrative Panel on Biosafety will request that the Principal Investigator indicate the source of any agents used in experiments at Stanford during the application process. If the investigator intends to obtain the agent from outside the United States, a copy of the CDC or other permit will be requested by the APB as part of the APB review of the application.
Items Requiring Permits
Etiologic agents
It is impractical to list all of the several hundred species of etiologic agents. In general, an import permit is needed for any infectious agent known to cause disease in man. This includes, but is not limited to, bacteria, viruses, rickettsia, parasites, yeasts, molds, and prions. In some instances, agents which are suspected of causing human disease also require a permit.
Biological materials
Unsterilized specimens of human and animal tissue (including blood), body discharges, fluids, excretions or similar material, when known or suspected of being infected with disease transmissible to humans require a permit under these provisions in order to be imported.
Animals
Any animal known or suspected of being infected with any disease transmissible to humans. Importation of turtles of less than 4 inches in shell length and all non-human primates requires an importation permit issued by the Division of Quarantine. Telephone 404-718-2077 for further information.
Insects
Any living insect, or other living arthropod, known or suspected of being infected with any disease transmissible to humans. Also, if alive, any fleas, flies, lice, mites, mosquitoes, or ticks, even if uninfected. This includes eggs, larvae, pupae, and nymphs as well as adult forms.
Snails
Any snails capable of transmitting schistosomiasis. No mollusks are to be admitted without a permit from either Centers for Disease Control or the Department of Agriculture. Any shipment of mollusks with a permit from either agency will be cleared immediately.
Bats
All live bats. Bats may also require a permit from the U.S. Department of Interior, Fish and Wildlife Services.
If you are not certain if the agents you intend to use require a CDC importation permit, please call the Biosafety Officer at 725.1473 for assistance in making the determination.
Additional information regarding CDC permitting:
Centers for Disease Control and Prevention
Import Permit Program
1600 Clifton Road NE, Mailstop A-46
Atlanta, GA 30333
Telephone: 404-718-2077
FAX: 404-471-8333
Email: importpermit@cdc.gov
Other Permits
United States Department of Agriculture (USDA) Animal and Plant Health Inspection Service (APHIS) permits are required for infectious agents of livestock and biological materials containing animal products, particularly livestock material.
Tissue (cell) culture techniques customarily use bovine material as a stimulant for cell growth. Tissue culture materials and suspensions of cell culture used to grow viruses or other etiologic agents and which contain growth stimulants of bovine or other livestock origin are, therefore, controlled by the USDA due to the potential risk of introduction of exotic animal diseases into the U.S. Further information may be obtained at https://www.aphis.usda.gov/aphis/resources/permits or by calling the USDA/APHIS at 1-888-272-3181.
United States Department of Interior (USDI) permits are required for certain live animals and all live bats. Go to https://www.fws.gov/service/permits?$skip=10 or call 800-358-2104 for further information.
Letters of Authorization
After a review of an “Application to Import an Etiological Agent”, the issuing officer may issue a “Letter of Authorization” rather than an importation permit. The Letter of Authorization is issued for materials that are judged to be non-infectious, but which might be construed to be infectious by U.S. Customs inspection personnel.
Letters of Authorization may be issued for items such as formalin fixed tissues, sterile cell cultures, clinical materials such as human blood, serum, plasma, urine, or cerebrospinal fluid, or other tissues or materials of human origin when there is no evidence or indication that such materials contain an infectious agent.
A copy of a Letter of Authorization should be attached to the package, and also should be furnished to the courier or importation broker. Letters of Authorization are in effect for two years.
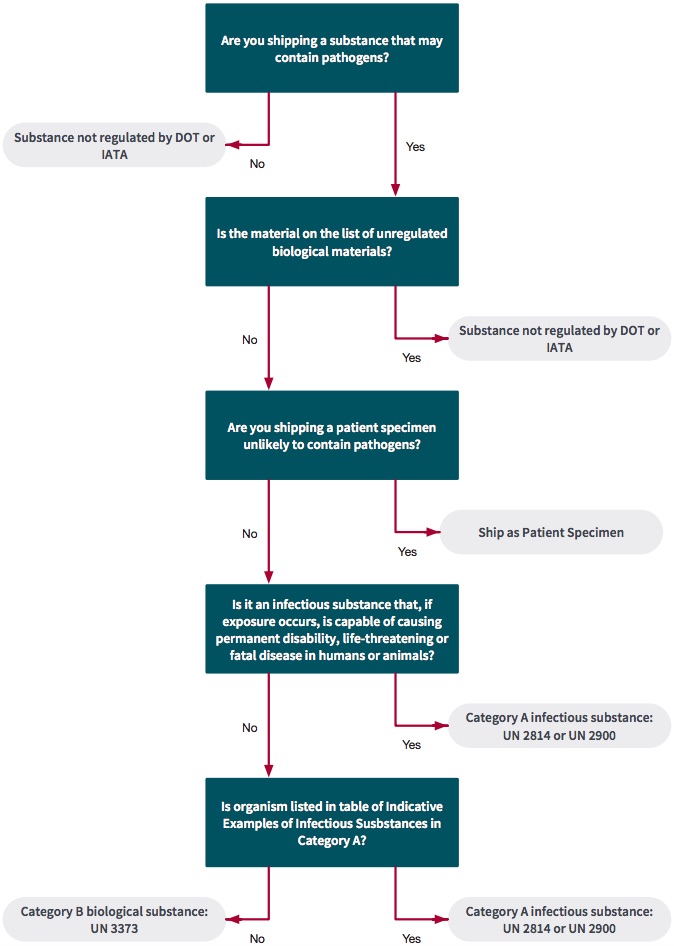
For training to become certified for shipping of biological goods, go to: https://ehs.stanford.edu/topic/biosafety-biosecurity/shipping
