Human Clinical Research Protocols (IRB/APB)
Protocols involving the use of infectious agents and/or rDNA/sNA for gene transfer into humans, whether done directly in the subject or in vitro and subsequently put into the subject, must be submitted to both the APB and the Stanford University Institutional Review Board (IRB) for Medical Human Subjects prior to initiation of protocol. The APB usually reviews the human clinical research protocols prior to the IRB review. Like the APB, the IRB uses eProtocol for its reviews.
Stem Cell Research (SCRO/APB)
All research involving the use of Stem Cells come under the Institutional Review Board/Stem Cell Research Oversight Panel (IRB/SCRO Panel) within the Research Compliance Office (RCO). As mandated by State Law, all University research projects involving human stem cells must be reviewed and approved by the IRB/SCRO Panel. The IRB/SCRO Panel is responsible for providing scientific and ethical review of all proposed research projects involving all human stem cells. This review is in addition to other compliance panel reviews that may be required such as Animal Care and Use, or Biosafety. The IRB/SCRO uses eProtocol for its reviews.
Research involving infectious agents and/or rDNA/sNA and Animals (APLAC/APB)
Research involving the use of infectious agents and/ or non-exempt rDNA/sNA with animals (in vivo) requires both APB and Administrative Panel on Laboratory Animal Care (APLAC) approval. These approvals are not required to be obtained in any specific sequence but both must be approved prior to any research being done. APLAC uses eProtocol for its reviews.
![]() Important Information
Important Information
Research Compliance Panels
at Stanford
| Panel | Oversight |
|---|---|
| APB | Biologicals BSL 2/3 rDNA/synthetic nucleic acids Prion/Prion-like proteins Use of biologicals/rDNA in humans Use of biologicals/rDNA in animals |
| APLAC | Animals |
| IRB | Human Subjects |
| SCRO | Human Stem Cells |
The APB Review Process
At Stanford University, research involving rDNA and/ or Biohazardous Agents is regulated by the NIH and comes under review of the Stanford University Administrative Panel on Biosafety (APB).
Research that involves the use of the above materials (excluding exempt rDNA (see Chapter 2) and clinical research that requires Institutional Biosafety Committee approval MUST be reviewed and approved by the APB prior to work being done. The APB, along with the Stanford IRBs, APLAC and SCRO, uses eProtocol, a web-based system that coordinates new protocols, updates, renewals, and reminders.
The flow chart in Figure 1 illustrates the eProtocol Biosafety workflow.
For information about eProtocol Biosafety, go to: https://eprotocol.stanford.edu/.
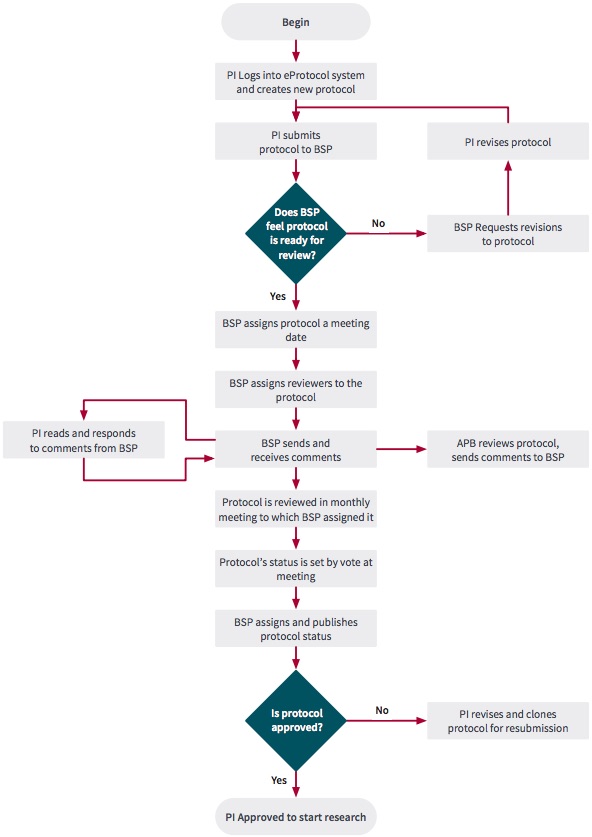
![]() Important Information
Important Information
APB Meeting Dates, Submission, and Protocol Cycle
The APB meets the third Wednesday of each month; protocol applications that require APB review, if submitted by the first of the month, will normally be reviewed by the panel at that month’s meeting.
Protocol Actions
Renewal: an update is required on the anniversary of the protocol approval; this is an opportunity to ensure that all personnel, project information, locations, etc. are current. The eProtocol system will send the PI, research coordinator and administrator (if designated) automatic email reminders prior to the due date. If an annual update is not submitted the eProtocol system will close the protocol.
Revision: any approved protocol can be revised at any time during its approval cycle to update personnel, project information, locations, etc. Revisions will be reviewed on the same panel schedual as noted above.
Protocol Terminations
Duration of approval: BSL – 2 projects are normally approved for three years, with a requirement for renewals (annual updates); BSL – 3 and human clinical projects are approved for one year. Annual updates and revisions of projects are done through eProtocol Biosafety.
Violations and Termination of APB Approval: An approved user who willfully or negligently violates the University, federal, or state rules and regulations governing the use of biohazardous agents/rDNA may have his/her APB approval suspended or revoked by the Biosafety Officer pending review by the APB. The Biosafety Officer will prepare a report which will describe the violations in detail and will discuss the matter with the Chair of the APB who will then determine the final course of action.
